Alzheimer's disease drug shelved after trial failure
- Published
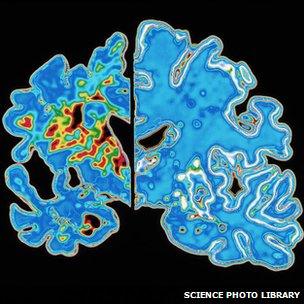
Alzheimer's significantly decreases brain volume (at left) in those affected
Two US drug firms say they will stop development of an Alzheimer's drug because it failed in two late-stage clinical trials.
Bapineuzumab, made by Pfizer and Johnson & Johnson, was designed to halt build-up of plaque in the brain.
But it failed to improve cognitive or functional performance compared with a placebo in certain patients.
Alzheimer's is the most common form of dementia, as well as the sixth leading cause of death in the United States.
An estimated 36 million people worldwide are believed to have dementia, including Alzheimer's.
Late stage trials
Both firms announced on 23 July that the first clinical trial of the intravenous (IV) version of bapineuzumab had failed.
In that study, patients with a gene that is associated with a greater risk of Alzheimer's were tested.
But results with the group were largely the same as with those who did not have the gene, who were tested in the second study.
The second trial's end means that additional studies on the IV version will not take place; however, Johnson and Johnson said a study of subcutaneous use would continue.
Some had predicted that the IV studies of bapineuzumab would fail because they were treating those whose brains were already damaged.
"One of the strong thoughts in the field is that you really have to treat people before they become demented," William Thies, chief scientific officer of the Alzheimer's Association, external told Reuters, adding that the announcement did not prevent the drug from being tested as a preventative.
And Mr Thies said that despite the trial's failure, data from the experiment could still be useful.
"These studies are terribly important for us to learn about Alzheimer's disease, and that part of the process is just starting as the data continues to be crunched in a variety of ways."
Johnson and Johnson had agreed in 2009 to invest up to $1.5bn (£961m) in bapineuzumab.
In a statement, Steven Romano, head of Pfizer's Medicines Development Group said they were "obviously very disappointed" with the trial's outcome.
"We are also saddened by the lost opportunity to provide a meaningful advance for patients afflicted with mild-to-moderate Alzheimer's disease and their caregivers," he said.
A similar drug being developed by Eli Lilly, solanezumab, is also considered a long-shot to succeed, but results of the trials will not be available until later this year.
- Published26 April 2012
- Published11 July 2012